Nestle violates law in India, conducts clinical trials on premature infants for baby food
In complete violation of the law, Nestle has conducted clinical trials on 75 premature babies in five hospitals on substitutes for breast milk

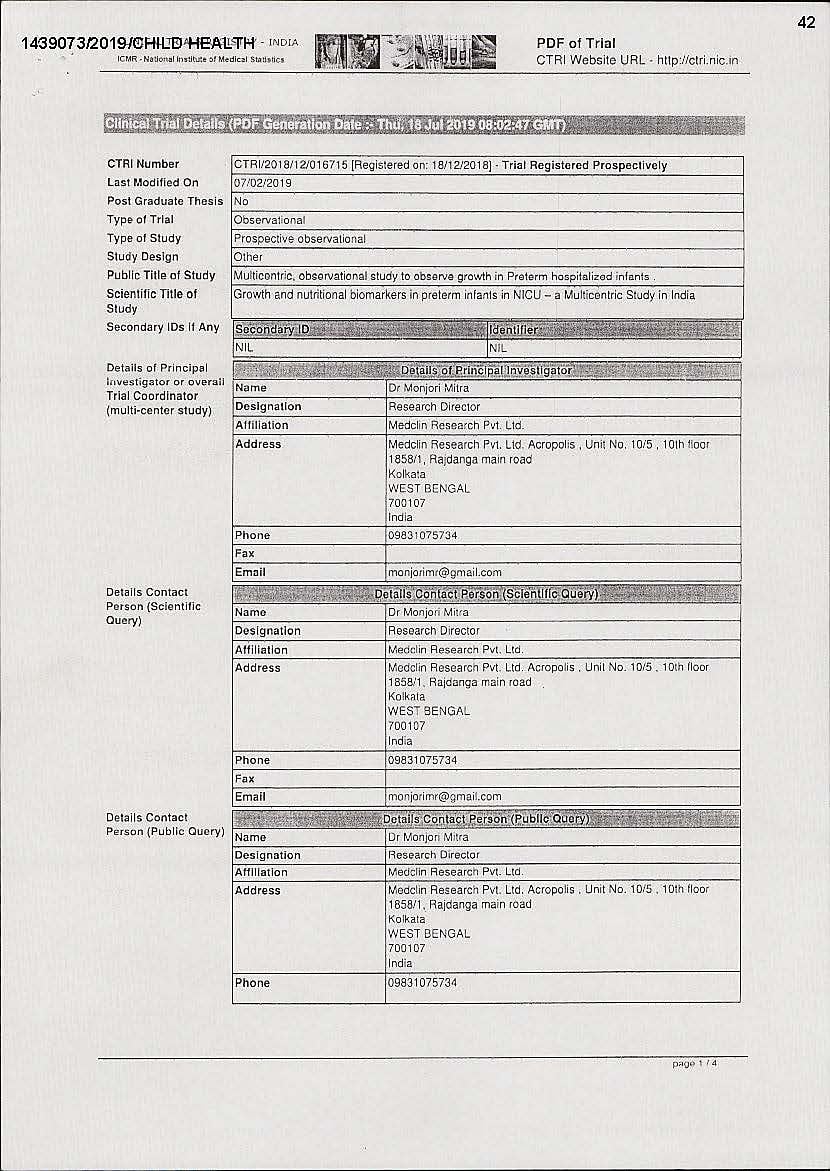
Breaking the law, Nestle has conducted clinical trials on 75 premature babies in five hospitals on substitutes for breast milk in complete contravention of the Infant Milk Substitutes Act. It was found by Breastfeeding Promotion Network of India (BPNI) on screening the trial registry of Indian Council of Medical Research (ICMR) that Nestle has sponsored a research titled “Multicentric Observational Study to Observe Growth in Preterm hospitalised infants”.
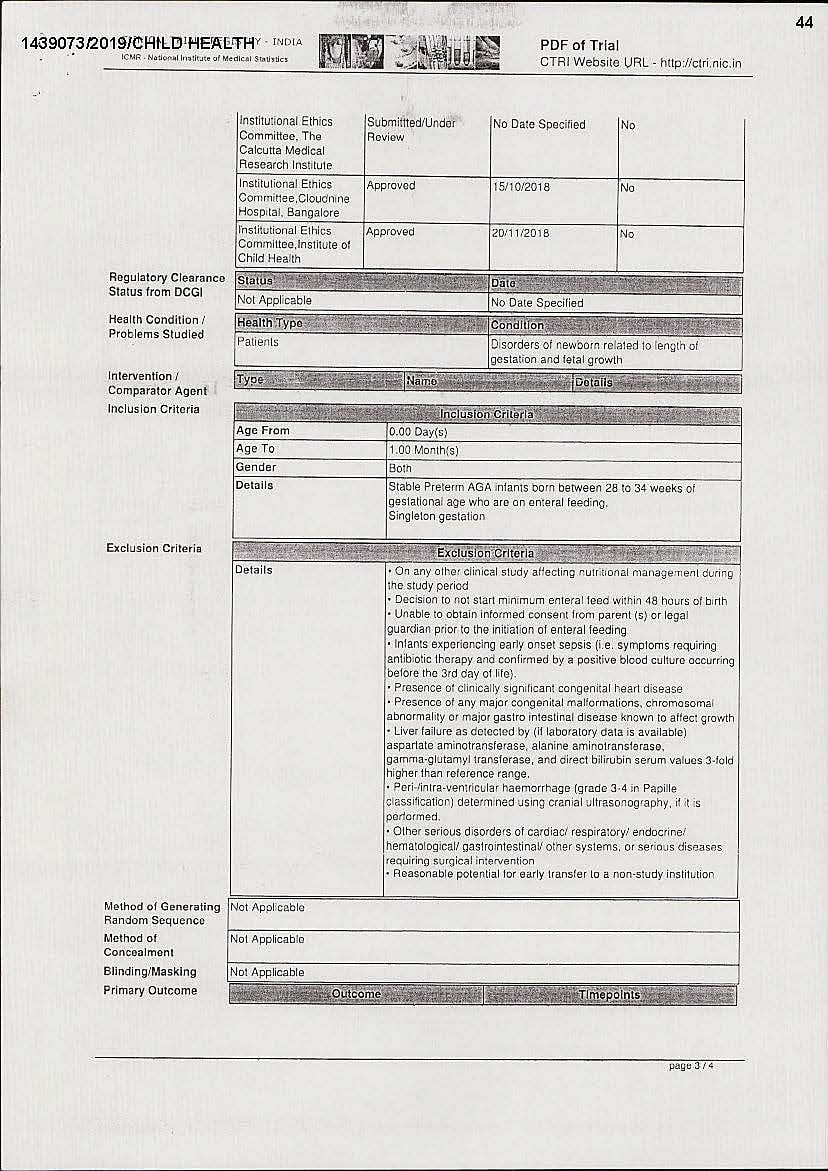
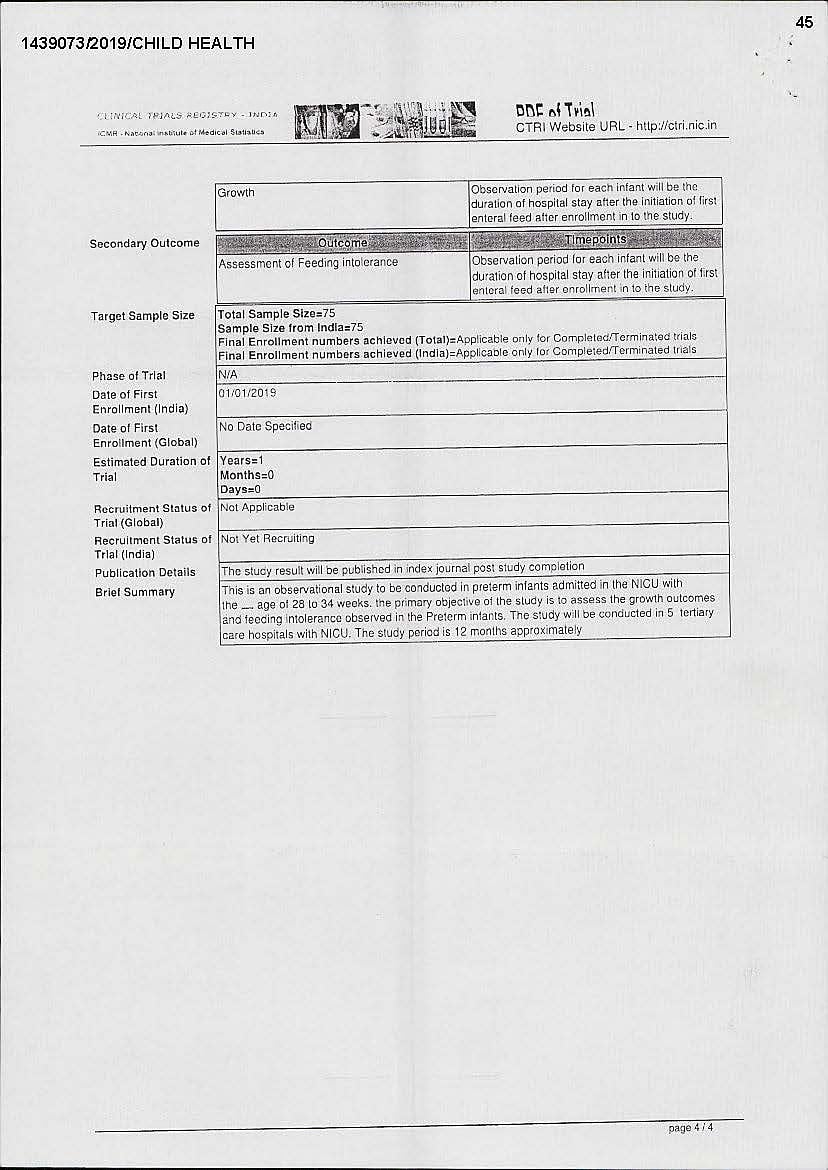
The trial was conducted on 75 premature babies between the age of 28-34 weeks. The objective of the study was to assess the growth and feeding intolerance in preterm infants. One of the exclusions was that the infant could not be fed milk substitute within 48 hours of birth, but from the third day onwards, the trial suggested that the infant could be given a milk substitute instead of breast milk.
BPNI complained to the ministry of health and family welfare and on receiving the complaint health secretary Preeti Sudan directed the ICMR director Balram Bhargava to monitor compliance of this clinical trial with law. Reprimanding ICMR for not having paid attention to their duty, Sudan directs them to ensure that all such clinical trials are first screened for infringement of the IMS Act.
Nestle violated Section 9(2) of the IMS Act. The Section states that no producer, supplier or distributor shall offer or give any contribution or pecuniary benefit to a health worker or any association of health workers, including funding of seminar, meeting, conferences, educational course, contest, fellowship, research work or sponsorship.
Section 9 of the IMS Act states that no person who produces, supplies, distributes or sells infant milk substitutes or feeding bottles or infant foods shall offer or give, directly or indirectly, any financial inducements or gifts to a health worker or to any member of his family for the purpose of promoting the use of such substitutes or bottles or foods.
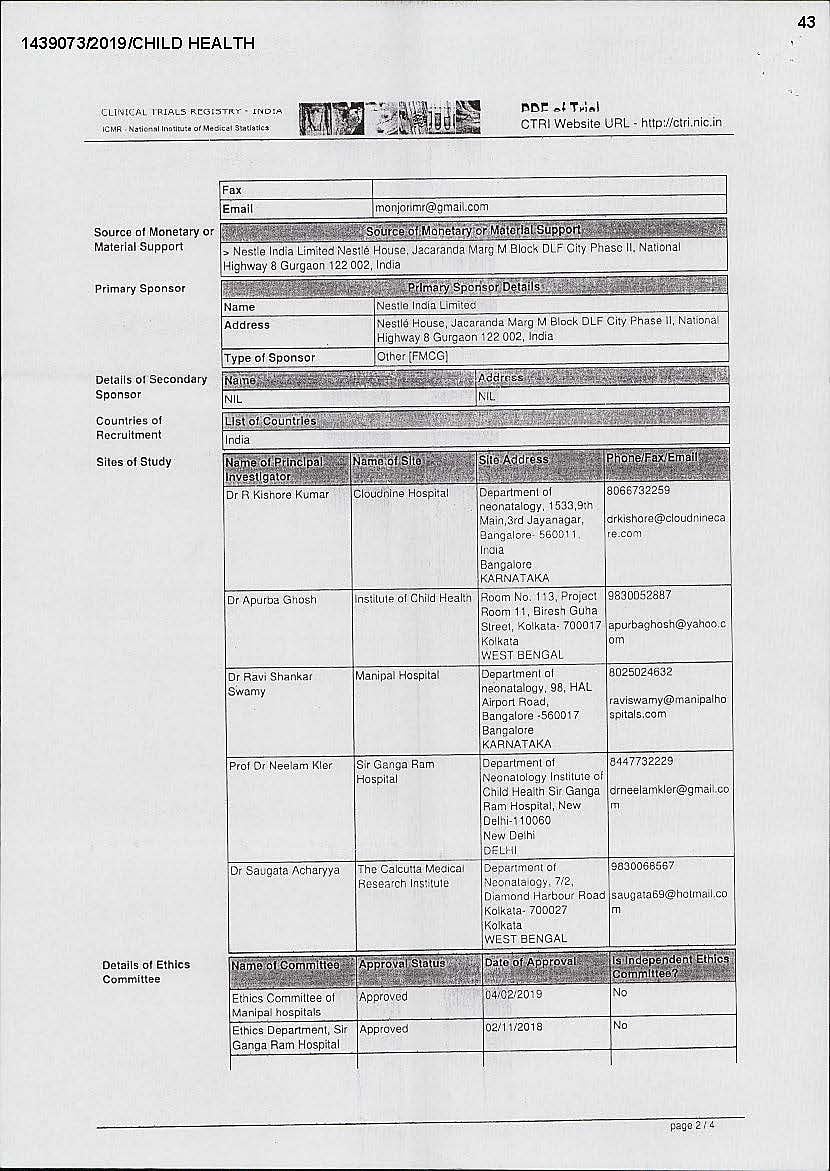
Dr Monjoni Mitra of Medclin Research is the trial co-ordinator and research director of the trial. The five hospitals which participated in the research — Cloudnine Hospital (Bengaluru), Institute of Child Health (Kolkata), Manipal Hospital (Bengaluru), Sir Ganga Ram Hospital (New Delhi) and Calcutta Medical Research Institute (Kolkata)— are private institutions. R Kishore Kumar, Apurba Ghosh, Ravi Shankar Swamy, Neelam Kher and Saugata Acharyya were the doctors involved in these respective hospitals for the trials.
“They are conducting these trials on premature babies. It is extremely risky. Babies are most sensitive during this period. What would have happened if anything went wrong with these babies,” asks Amulya Nidhi, co-convenor of Swasth Adhikar Manch.
While conducting such clinical trials, the approval of the Drug Controller General of India (DCGI) is required. “In the application, Nestle mentions that regulatory clearance from DCGI is not required. So, who then monitors such trials? Also, the application does not disclose who all are there in the ethics committee within these hospitals to approve such trials. Are there paediatricians, nutritional experts and what expertise do they have for approving such trials. In fact, the trial was conducted at Calcutta Medical Research Institute even before the in-house ethics committee approved the trial,” underscored Nidhi.
“The Ministry has directed ICMR to regulate a trial that is already underway. The question is how are such trials happening under the watch of ICMR? You can’t regulate a trial retrospectively. It has to be done before the trials begin,” explained Dr Sylvia Karpagam, public health doctor and researcher.
“This is not the first time that Nestle has tried to enter the market. They don’t follow protocols. They have been trying to bring in un-regulated baby food into the market. This research has not even been approved by an independent ethics committee. Nestle is violating the IMS Act. They should adhere to the law. We are talking about food for infants,” observes Karpagam.
“When such trials are held, doctors get monetary benefits. They begin to promote powder food for babies even when it is not necessary. They keep telling the mother that the baby is not being fed well. There is no need for any other liquid or powder for a baby till six months,” says Karpagam.
Unregulated clinical trials are a norm in India. “In a report by PricewaterhouseCoopers on emerging markets in India, it had stated that the country is a good ground for clinical trial,” highlighted Karpagam.
According to a Supreme Court ruling, clinical trials can be approved in India only in three instances. If it is related to an unmet medical need of the country; In case of innovations vs existing therapy and on basis of the risk benefit analysis.
“Nestle’s clinical trials don’t fall under any of these categories. It is not a ‘need’. Here, premature babies are being put at risk. The country spends crores to improve infant mortality rates and here babies are put at risk with this trial. Moreover, Nestle has only commercial interests on its mind,” insists Nidhi.
ICMR History with clinical trials
Since 2007, ICMR was part of clinical trials for administration of anti-cervical cancer HPV vaccines to prevent cervical cancer in mong 24,000 girls in the tribal belts of Andhra Pradesh and Gujarat. In 2010, six tribal girls from Gujarat and Andhra Pradesh involved these trials died.
The vaccines, produced by Merck and GlaxoSmithKline, were given under an “observational study” conducted under the aegis of Path (Programme for Appropriate Technology in Health), an organisation funded by the Bill and Melinda Gates Foundation. These programmes were conducted in 2007 while the vaccines were approved later.
Under rules, a firm interested in trial is supposed to approach the Drug Controller with a protocol to get approvals, but in India there have been several evidences of weak monitoring.
The government had in 2010 stated that 1,725 persons have lost their lives to drug trials in four years from 2007 to 2010.
A Parliamentary committee report in 2010 found that ICMR had signed a memorandum of understanding to provide technical support to the project in 2006 even before the Drugs Controller General of India (DGCI) approved its use in the country, which actually happened in 2008. An ICMR official was implicated in this for favouring this project.
Follow us on: Facebook, Twitter, Google News, Instagram
Join our official telegram channel (@nationalherald) and stay updated with the latest headlines