Can India's new cervical cancer vaccine protect more women?
According to the World Health Organization (WHO), 4.1 million women in India have died from the disease since 2019, and without intervention, as many as 5.7 million will die by 2070
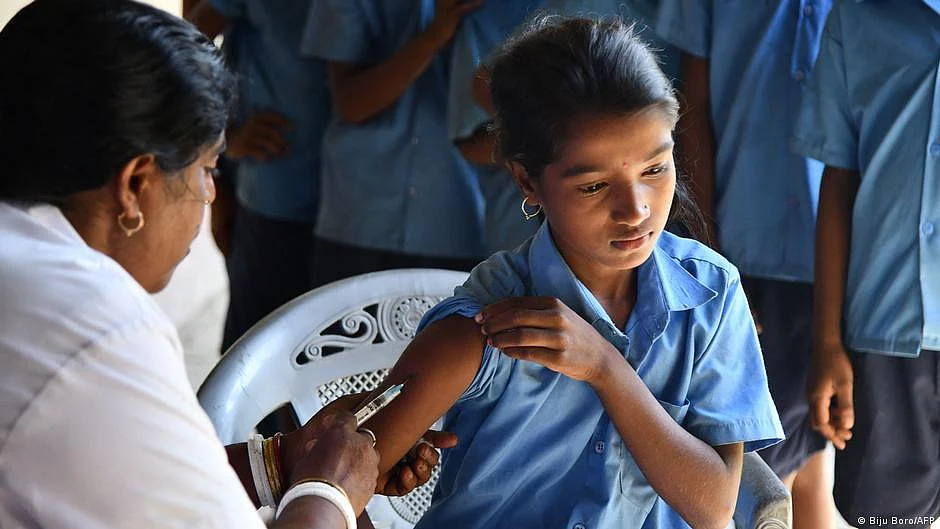
The Indian government recently announced that it will begin a nationwide immunization drive in April for girls aged between 9 and 14 using a newly developed vaccine targeting human papilloma virus (HPV), the main cause of cervical cancer and a potential cause of other cancers.
According to the World Health Organization (WHO), 4.1 million women in India have died from the disease since 2019, and without intervention, as many as 5.7 million will die by 2070.
The WHO has specified that countries should reach and maintain an incidence rate of fewer than 4 new cases per 100,000 women a year by 2030. Preventative vaccination of girls is therefore essential to lowering the incidence rate.
"We have prepared a proper roadmap and a social mobilization strategy for the vaccine rollout. Our experience with the COVID vaccine program has helped us immensely," N K Arora, chairperson of the National Technical Advisory Group on Immunization, told DW.
The quadrivalent vaccine, called "Cervavac," developed by the Serum Institute of India (SII), offers protection against four strains of HPV.
At present, India is fully dependent on foreign manufacturers for the vaccines, which are expensive.
Following positive data from a large-phase clinical trial, marketing authorization was granted by the Drugs Controller General of India in July 2022.
India seeks more affordable immunization against HPV
HPV is a sexually transmitted viral infection, which affects the genitals, mouth and throat. Other than treating symptoms, there is no treatment for the virus itself, and prevention is key.
Although HPV vaccines have been available in India since 2008, attempts to establish a national vaccination program have stalled, mainly due to unfounded worries about side-effects, as well as affordability concerns, according to the medical journal, Lancet.
Production of Ceravac began at the end of 2022. It will be made available at a price of 200 to 400 Indian rupees ($2.45-$4.90, €2.25-€4.55) per dose.
SII is expected to produce around 200 million doses in about two years. Physical trials for the vaccine were held in ten health centers across India.
"First, it will be extended to citizens of India, and later the vaccine will go global. We have made it for all women around the world. It is very important for women's health and safety," Adar Poonawalla, CEO of SII, told DW.
The Serum Institute is the world's largest vaccine manufacturer by volume of doses it has produced and sold globally. It makes vaccines against diphtheria, tetanus, pertussis, hepatitis B, measles, mumps and rubella, and ships out more than 1.5 billion vaccine doses annually at affordable prices.
It is estimated that about 65% of the children in the world receive at least one vaccine manufactured by SII. Its vaccines are used in around 170 countries for national immunization programs, saving millions of lives throughout the world.
Community prevention of cervical cancer
Public health experts point out that screening is the core pillar of cervical cancer control. Well-organized and accessible national screening programs are essential.
Given that fewer that only 10% of women in India have been screened for cervical cancer in the past 5 years, much remains to be done.
"At primary and community health centers, staff nurses and medical officers should receive training for screening cervical cancer," Sarojini Nadimpally, public health researcher, told DW.
"It is important to look into health care infrastructure, including human resources at these centers. They are lacking in potential staffing that could be trained to carry out the screening," she said.
"For any vaccine, long-term monitoring is important. In the case of Cervavac, which is a two-dose program, it is important to understand the monitoring mechanism," she added.
The government has clarified that girls who do not attend schools will be given the vaccines by community outreach and mobile health teams.
A recent state-wide health screening program revealed that for every 100 women examined, one was detected with undiagnosed cervical cancer in the western state of Maharashtra.
In 2020, the northern state of Uttar Pradesh recorded 10,046 cases, making it the state with the highest estimated incidence of cervical cancer.
The Indian vaccine is expected to be a game changer that would bring down the incidence of cervical cancer because of its much lower cost.
"Partnering the strategy though public-private partnerships can expand the coverage to around 50 million girls of the age group 9-14 years. Generating awareness regarding the disease and vaccine can increase the acceptability of the vaccine," Giridhara Babu, an epidemiologist at the Public Health Foundation of India, told DW.
"Coupled with this, we need measures such as increased cancer screening at a very early stage," Babu added.
India has a robust vaccine distribution infrastructure. An "electronic vaccine intelligence network" enhances management of vaccine supplies, logistics and temperature tracking at all levels of vaccine storage from the national to the district level.
"In remote and inaccessible places, intensive outreach vaccination campaigns can be integrated into national health missions," said Babu.
The WHO has specified that countries should reach and maintain an incidence rate of fewer than 4 new cases per 100,000 women a year by 2030.
Edited by: Wesley Rahn
Follow us on: Facebook, Twitter, Google News, Instagram, WhatsApp
Join our official telegram channel (@nationalherald) and stay updated with the latest headlines
