Antiviral drugs may reduce monkeypox symptoms, shorten contagious phase: Lancet study
As optimum infection control and treatment strategies for this disease are not yet established, data from the study could help understand clinical features of the disease and transmission dynamics
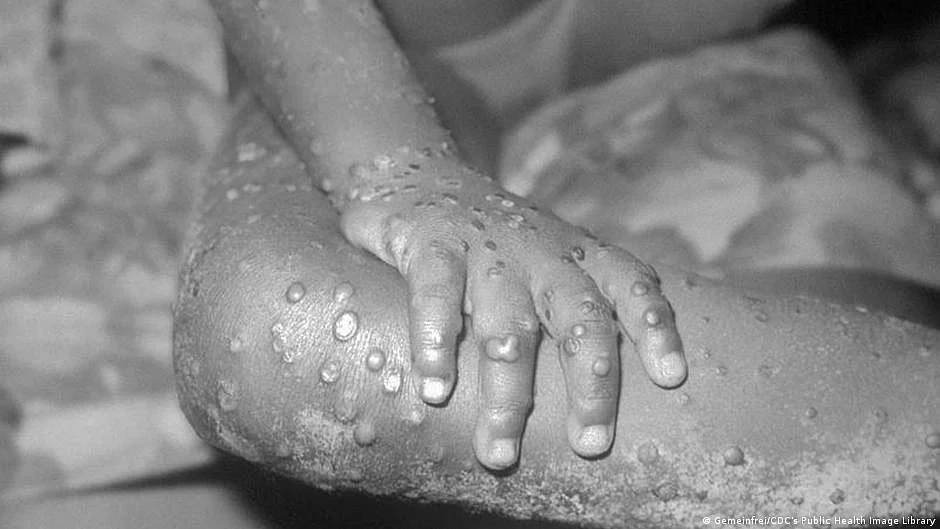
Some antiviral medications might have the potential to shorten symptoms of monkeypox and reduce the amount of time a patient is contagious, according to a study of seven patients diagnosed with the rare viral disease in the UK between 2018 and 2021.
The cases analysed in the study, published on Tuesday in The Lancet Infectious Diseases journal, represent the first instances of in-hospital transmission and household transmission outside of Africa.
The research also reported the patient response to the first off-label use of two different antiviral medications -- brincidofovir and tecovirimat -- to treat the disease.
The study found little evidence that brincidofovir was of clinical benefit but concluded that further research into the potential of tecovirimat would be warranted.
The researchers also report detection of monkeypox virus in blood and throat swabs.
As optimum infection control and treatment strategies for this disease are not yet established, data from the study could help inform global efforts to further understand the clinical features of the disease as well as transmission dynamics, they said.
"As public health officials are trying to understand what is causing the May 2022 monkeypox outbreaks in Europe and North America -- which have affected several patients who reported neither travel nor an identified link to a previously known case -- our study offers some of the first insights into the use of antivirals for the treatment of monkeypox in humans," said Hugh Adler of the Liverpool University Hospitals NHS Foundation Trust, UK.
"Although this latest outbreak has affected more patients than we had previously encountered in the UK, historically monkeypox has not transmitted very efficiently between people, and overall the risk to public health is low," Adler, lead author on the paper, said.
With international travel returning to pre-pandemic levels, public health officials and healthcare workers around the world must remain vigilant to the possibility of new cases of monkeypox, said Nick Price of Guy's & St Thomas' NHS Foundation Trust, senior author on the paper.
Of the seven UK monkeypox cases analysed in the study, four were imported from West Africa with three further cases occurring due to human-to-human transmission within the case clusters.
Monkeypox, a close relative of the smallpox virus, is a rare disease classified as a High Consequence Infectious Disease (HCID) by the UK Health Security Agency.
There are currently no licensed treatments for monkeypox and there is limited data on the duration of its contagiousness, with the incubation period ranging from five to 21 days.
The study authors observed clinical data alongside laboratory results from blood tests and nose-and-throat swabs to describe the duration and clinical features of monkeypox in a high-income setting.
Researchers also reported patient response to antiviral medications developed to treat smallpox -- brincidofovir and tecovirimat -- which have previously demonstrated some efficacy against monkeypox in animals.
Between 2018 and 2019, four patients observed in the study were treated for monkeypox in HCID units in England. Three of these cases were imported from West Africa.
The fourth case occurred in a healthcare worker 18 days after initial exposure to the virus and was the first example of monkeypox transmission in a hospital setting outside of Africa.
The initial three patients were treated with brincidofovir seven days after the initial onset of the rash.
The study found that brincidofovir did not have any convincing clinical benefit in treating monkeypox and alterations in liver blood tests were observed.
The researchers noted that it is not known whether brincidofovir administration earlier in the course of the disease or at a different dosing schedule would have yielded different clinical outcomes.
Despite this, all three patients, plus the fourth hospital transmission patient, made a full recovery, they said.
Three further cases of monkeypox were reported in the UK in 2021 in a family travelling from Nigeria, with two of these cases being the first examples of household transmission outside of Africa.
One of the 2021 UK patients was treated with tecovirimat and experienced a shorter duration of symptoms and upper respiratory tract viral shedding than the other cases in this cluster, the researchers said.
However, they note that conclusions are unable to be drawn on antiviral effectiveness against monkeypox in such a small cohort, calling for further research into antivirals to treat this neglected tropical disease.
"During previous outbreaks of monkeypox, patients were considered infectious until all lesions crusted over," said Catherine Houlihan, of the UK Health Security Agency and University College London, one of the co-authors of the paper.
"In these seven UK cases, viral shedding was observed for at least three weeks following infection. However, data on infectivity remains limited, and is an important area for future study," said Houlihan.
The authors acknowledge some limitations with this study, primarily its observational nature and the small number of cases analysed.
They were also unable to confirm positive monkeypox PCR test results with laboratory samples of the virus, meaning that ongoing shedding of contagious virus was unable to be verified.
Follow us on: Facebook, Twitter, Google News, Instagram, WhatsApp
Join our official telegram channel (@nationalherald) and stay updated with the latest headlines
